Seznamy 91 Classical Solar System Atomic Model
Seznamy 91 Classical Solar System Atomic Model. He realized that certain colors of light were given off when elements were exposed to flame or electric fields. Neils bohr came up the solar system model of the atom in 1913. He was a danish scientist who is best known for his contributions to the atomic model.
Prezentováno Amazon Com Lamvpker Solar System Dimple Simple Planet Dimple Oversize Poper Fidget Space Astronomy Space Toys Educational Toys For Party Favors Birthday Gift Toys Games
It shows that the negatively charged electron is attracted to the positively charged proton and so eventually the electron revolving around the nucleus is pulled into the center and the wholething will explode. He realized that certain colors of light were given off when elements were exposed to flame or electric fields. This picture was called the planetary model, since it pictured the atom as a miniature "solar system" with the electrons orbiting the nucleus.It shows that the negatively charged electron is attracted to the positively charged proton and so eventually the electron revolving around the nucleus is pulled into the center and the wholething will explode.
This picture was called the planetary model, since it pictured the atom as a miniature "solar system" with the electrons orbiting the nucleus. He realized that certain colors of light were given off when elements were exposed to flame or electric fields. The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … He was a danish scientist who is best known for his contributions to the atomic model. Neils bohr came up the solar system model of the atom in 1913. He was the first to realize that electrons travel in separate orbits around the nucleus. Following the work of ernest rutherford and his colleagues in the early twentieth century, the picture of atoms consisting of tiny dense nuclei surrounded by lighter and even tinier electrons continually moving about the nucleus was well established. The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets).
Following the work of ernest rutherford and his colleagues in the early twentieth century, the picture of atoms consisting of tiny dense nuclei surrounded by lighter and even tinier electrons continually moving about the nucleus was well established. He realized that certain colors of light were given off when elements were exposed to flame or electric fields. Neils bohr came up the solar system model of the atom in 1913. The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus …. This picture was called the planetary model, since it pictured the atom as a miniature "solar system" with the electrons orbiting the nucleus.

Neils bohr came up the solar system model of the atom in 1913. This picture was called the planetary model, since it pictured the atom as a miniature "solar system" with the electrons orbiting the nucleus. It was basically a modified version of rutherford's atomic model wherein bohr explained that electrons move in fixed orbitals (shells) and not anywhere in between and he also explained that each orbit (shell) has a …. He was a danish scientist who is best known for his contributions to the atomic model.

The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … He realized that certain colors of light were given off when elements were exposed to flame or electric fields. This picture was called the planetary model, since it pictured the atom as a miniature "solar system" with the electrons orbiting the nucleus. The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets). He was a danish scientist who is best known for his contributions to the atomic model. Neils bohr came up the solar system model of the atom in 1913. He was the first to realize that electrons travel in separate orbits around the nucleus... It shows that the negatively charged electron is attracted to the positively charged proton and so eventually the electron revolving around the nucleus is pulled into the center and the wholething will explode.

Following the work of ernest rutherford and his colleagues in the early twentieth century, the picture of atoms consisting of tiny dense nuclei surrounded by lighter and even tinier electrons continually moving about the nucleus was well established. He was the first to realize that electrons travel in separate orbits around the nucleus. The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets). He realized that certain colors of light were given off when elements were exposed to flame or electric fields. He was a danish scientist who is best known for his contributions to the atomic model. Neils bohr came up the solar system model of the atom in 1913. It shows that the negatively charged electron is attracted to the positively charged proton and so eventually the electron revolving around the nucleus is pulled into the center and the wholething will explode. It was basically a modified version of rutherford's atomic model wherein bohr explained that electrons move in fixed orbitals (shells) and not anywhere in between and he also explained that each orbit (shell) has a … Following the work of ernest rutherford and his colleagues in the early twentieth century, the picture of atoms consisting of tiny dense nuclei surrounded by lighter and even tinier electrons continually moving about the nucleus was well established. The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … This picture was called the planetary model, since it pictured the atom as a miniature "solar system" with the electrons orbiting the nucleus. Following the work of ernest rutherford and his colleagues in the early twentieth century, the picture of atoms consisting of tiny dense nuclei surrounded by lighter and even tinier electrons continually moving about the nucleus was well established.

It shows that the negatively charged electron is attracted to the positively charged proton and so eventually the electron revolving around the nucleus is pulled into the center and the wholething will explode... The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … Neils bohr came up the solar system model of the atom in 1913. He was the first to realize that electrons travel in separate orbits around the nucleus.. He was the first to realize that electrons travel in separate orbits around the nucleus.

He was a danish scientist who is best known for his contributions to the atomic model.. It shows that the negatively charged electron is attracted to the positively charged proton and so eventually the electron revolving around the nucleus is pulled into the center and the wholething will explode.
This picture was called the planetary model, since it pictured the atom as a miniature "solar system" with the electrons orbiting the nucleus. Neils bohr came up the solar system model of the atom in 1913. The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … Following the work of ernest rutherford and his colleagues in the early twentieth century, the picture of atoms consisting of tiny dense nuclei surrounded by lighter and even tinier electrons continually moving about the nucleus was well established. He realized that certain colors of light were given off when elements were exposed to flame or electric fields. He realized that certain colors of light were given off when elements were exposed to flame or electric fields.

He was a danish scientist who is best known for his contributions to the atomic model. The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus ….. It was basically a modified version of rutherford's atomic model wherein bohr explained that electrons move in fixed orbitals (shells) and not anywhere in between and he also explained that each orbit (shell) has a …
He realized that certain colors of light were given off when elements were exposed to flame or electric fields. He realized that certain colors of light were given off when elements were exposed to flame or electric fields. It was basically a modified version of rutherford's atomic model wherein bohr explained that electrons move in fixed orbitals (shells) and not anywhere in between and he also explained that each orbit (shell) has a … Neils bohr came up the solar system model of the atom in 1913. It shows that the negatively charged electron is attracted to the positively charged proton and so eventually the electron revolving around the nucleus is pulled into the center and the wholething will explode. He was the first to realize that electrons travel in separate orbits around the nucleus. This picture was called the planetary model, since it pictured the atom as a miniature "solar system" with the electrons orbiting the nucleus. The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … He was a danish scientist who is best known for his contributions to the atomic model.. This picture was called the planetary model, since it pictured the atom as a miniature "solar system" with the electrons orbiting the nucleus.

Following the work of ernest rutherford and his colleagues in the early twentieth century, the picture of atoms consisting of tiny dense nuclei surrounded by lighter and even tinier electrons continually moving about the nucleus was well established.. It shows that the negatively charged electron is attracted to the positively charged proton and so eventually the electron revolving around the nucleus is pulled into the center and the wholething will explode. He was the first to realize that electrons travel in separate orbits around the nucleus. It was basically a modified version of rutherford's atomic model wherein bohr explained that electrons move in fixed orbitals (shells) and not anywhere in between and he also explained that each orbit (shell) has a … The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets). The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus ….. He realized that certain colors of light were given off when elements were exposed to flame or electric fields.

This picture was called the planetary model, since it pictured the atom as a miniature "solar system" with the electrons orbiting the nucleus. He realized that certain colors of light were given off when elements were exposed to flame or electric fields. Neils bohr came up the solar system model of the atom in 1913. The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets)... Following the work of ernest rutherford and his colleagues in the early twentieth century, the picture of atoms consisting of tiny dense nuclei surrounded by lighter and even tinier electrons continually moving about the nucleus was well established.

Neils bohr came up the solar system model of the atom in 1913. The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets). He was a danish scientist who is best known for his contributions to the atomic model. The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … It shows that the negatively charged electron is attracted to the positively charged proton and so eventually the electron revolving around the nucleus is pulled into the center and the wholething will explode. He realized that certain colors of light were given off when elements were exposed to flame or electric fields. Following the work of ernest rutherford and his colleagues in the early twentieth century, the picture of atoms consisting of tiny dense nuclei surrounded by lighter and even tinier electrons continually moving about the nucleus was well established. He was the first to realize that electrons travel in separate orbits around the nucleus.

The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets)... This picture was called the planetary model, since it pictured the atom as a miniature "solar system" with the electrons orbiting the nucleus. It was basically a modified version of rutherford's atomic model wherein bohr explained that electrons move in fixed orbitals (shells) and not anywhere in between and he also explained that each orbit (shell) has a … Neils bohr came up the solar system model of the atom in 1913. The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … He was the first to realize that electrons travel in separate orbits around the nucleus. The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets). He realized that certain colors of light were given off when elements were exposed to flame or electric fields. The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets).
He was a danish scientist who is best known for his contributions to the atomic model... The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets). This picture was called the planetary model, since it pictured the atom as a miniature "solar system" with the electrons orbiting the nucleus. Neils bohr came up the solar system model of the atom in 1913. The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … It was basically a modified version of rutherford's atomic model wherein bohr explained that electrons move in fixed orbitals (shells) and not anywhere in between and he also explained that each orbit (shell) has a …

It shows that the negatively charged electron is attracted to the positively charged proton and so eventually the electron revolving around the nucleus is pulled into the center and the wholething will explode. Neils bohr came up the solar system model of the atom in 1913. It was basically a modified version of rutherford's atomic model wherein bohr explained that electrons move in fixed orbitals (shells) and not anywhere in between and he also explained that each orbit (shell) has a … He was the first to realize that electrons travel in separate orbits around the nucleus.. It was basically a modified version of rutherford's atomic model wherein bohr explained that electrons move in fixed orbitals (shells) and not anywhere in between and he also explained that each orbit (shell) has a …

The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets)... He was the first to realize that electrons travel in separate orbits around the nucleus. Following the work of ernest rutherford and his colleagues in the early twentieth century, the picture of atoms consisting of tiny dense nuclei surrounded by lighter and even tinier electrons continually moving about the nucleus was well established. It shows that the negatively charged electron is attracted to the positively charged proton and so eventually the electron revolving around the nucleus is pulled into the center and the wholething will explode... This picture was called the planetary model, since it pictured the atom as a miniature "solar system" with the electrons orbiting the nucleus.

The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets). This picture was called the planetary model, since it pictured the atom as a miniature "solar system" with the electrons orbiting the nucleus... He realized that certain colors of light were given off when elements were exposed to flame or electric fields.

He was the first to realize that electrons travel in separate orbits around the nucleus. He was the first to realize that electrons travel in separate orbits around the nucleus. The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … This picture was called the planetary model, since it pictured the atom as a miniature "solar system" with the electrons orbiting the nucleus.. Neils bohr came up the solar system model of the atom in 1913.

He was the first to realize that electrons travel in separate orbits around the nucleus.. Following the work of ernest rutherford and his colleagues in the early twentieth century, the picture of atoms consisting of tiny dense nuclei surrounded by lighter and even tinier electrons continually moving about the nucleus was well established. He was the first to realize that electrons travel in separate orbits around the nucleus. Neils bohr came up the solar system model of the atom in 1913. He was a danish scientist who is best known for his contributions to the atomic model. It was basically a modified version of rutherford's atomic model wherein bohr explained that electrons move in fixed orbitals (shells) and not anywhere in between and he also explained that each orbit (shell) has a … It shows that the negatively charged electron is attracted to the positively charged proton and so eventually the electron revolving around the nucleus is pulled into the center and the wholething will explode.. It shows that the negatively charged electron is attracted to the positively charged proton and so eventually the electron revolving around the nucleus is pulled into the center and the wholething will explode.

He was a danish scientist who is best known for his contributions to the atomic model. He realized that certain colors of light were given off when elements were exposed to flame or electric fields. He was a danish scientist who is best known for his contributions to the atomic model. Neils bohr came up the solar system model of the atom in 1913. The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets). The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … He was the first to realize that electrons travel in separate orbits around the nucleus. It was basically a modified version of rutherford's atomic model wherein bohr explained that electrons move in fixed orbitals (shells) and not anywhere in between and he also explained that each orbit (shell) has a … Following the work of ernest rutherford and his colleagues in the early twentieth century, the picture of atoms consisting of tiny dense nuclei surrounded by lighter and even tinier electrons continually moving about the nucleus was well established.. The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus …

It was basically a modified version of rutherford's atomic model wherein bohr explained that electrons move in fixed orbitals (shells) and not anywhere in between and he also explained that each orbit (shell) has a ….. He was a danish scientist who is best known for his contributions to the atomic model. Neils bohr came up the solar system model of the atom in 1913. The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … Following the work of ernest rutherford and his colleagues in the early twentieth century, the picture of atoms consisting of tiny dense nuclei surrounded by lighter and even tinier electrons continually moving about the nucleus was well established. It shows that the negatively charged electron is attracted to the positively charged proton and so eventually the electron revolving around the nucleus is pulled into the center and the wholething will explode. The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets). He was the first to realize that electrons travel in separate orbits around the nucleus. This picture was called the planetary model, since it pictured the atom as a miniature "solar system" with the electrons orbiting the nucleus.. He was the first to realize that electrons travel in separate orbits around the nucleus.
Neils bohr came up the solar system model of the atom in 1913. It was basically a modified version of rutherford's atomic model wherein bohr explained that electrons move in fixed orbitals (shells) and not anywhere in between and he also explained that each orbit (shell) has a … The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets). The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus …

The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … He was the first to realize that electrons travel in separate orbits around the nucleus. It was basically a modified version of rutherford's atomic model wherein bohr explained that electrons move in fixed orbitals (shells) and not anywhere in between and he also explained that each orbit (shell) has a …. He realized that certain colors of light were given off when elements were exposed to flame or electric fields.
Neils bohr came up the solar system model of the atom in 1913. He was the first to realize that electrons travel in separate orbits around the nucleus. The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … He realized that certain colors of light were given off when elements were exposed to flame or electric fields. It shows that the negatively charged electron is attracted to the positively charged proton and so eventually the electron revolving around the nucleus is pulled into the center and the wholething will explode. Following the work of ernest rutherford and his colleagues in the early twentieth century, the picture of atoms consisting of tiny dense nuclei surrounded by lighter and even tinier electrons continually moving about the nucleus was well established. Neils bohr came up the solar system model of the atom in 1913. He was a danish scientist who is best known for his contributions to the atomic model.. Neils bohr came up the solar system model of the atom in 1913.

The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets). It was basically a modified version of rutherford's atomic model wherein bohr explained that electrons move in fixed orbitals (shells) and not anywhere in between and he also explained that each orbit (shell) has a … It shows that the negatively charged electron is attracted to the positively charged proton and so eventually the electron revolving around the nucleus is pulled into the center and the wholething will explode. Neils bohr came up the solar system model of the atom in 1913.

He realized that certain colors of light were given off when elements were exposed to flame or electric fields. The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … He was the first to realize that electrons travel in separate orbits around the nucleus. This picture was called the planetary model, since it pictured the atom as a miniature "solar system" with the electrons orbiting the nucleus. Neils bohr came up the solar system model of the atom in 1913. The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets). Following the work of ernest rutherford and his colleagues in the early twentieth century, the picture of atoms consisting of tiny dense nuclei surrounded by lighter and even tinier electrons continually moving about the nucleus was well established. He was a danish scientist who is best known for his contributions to the atomic model. It shows that the negatively charged electron is attracted to the positively charged proton and so eventually the electron revolving around the nucleus is pulled into the center and the wholething will explode. He realized that certain colors of light were given off when elements were exposed to flame or electric fields. It was basically a modified version of rutherford's atomic model wherein bohr explained that electrons move in fixed orbitals (shells) and not anywhere in between and he also explained that each orbit (shell) has a …. He was a danish scientist who is best known for his contributions to the atomic model.

He was the first to realize that electrons travel in separate orbits around the nucleus. The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets). Neils bohr came up the solar system model of the atom in 1913. It shows that the negatively charged electron is attracted to the positively charged proton and so eventually the electron revolving around the nucleus is pulled into the center and the wholething will explode. Following the work of ernest rutherford and his colleagues in the early twentieth century, the picture of atoms consisting of tiny dense nuclei surrounded by lighter and even tinier electrons continually moving about the nucleus was well established. He was the first to realize that electrons travel in separate orbits around the nucleus. He was a danish scientist who is best known for his contributions to the atomic model. This picture was called the planetary model, since it pictured the atom as a miniature "solar system" with the electrons orbiting the nucleus. It was basically a modified version of rutherford's atomic model wherein bohr explained that electrons move in fixed orbitals (shells) and not anywhere in between and he also explained that each orbit (shell) has a … He realized that certain colors of light were given off when elements were exposed to flame or electric fields.. This picture was called the planetary model, since it pictured the atom as a miniature "solar system" with the electrons orbiting the nucleus.

It shows that the negatively charged electron is attracted to the positively charged proton and so eventually the electron revolving around the nucleus is pulled into the center and the wholething will explode.. He realized that certain colors of light were given off when elements were exposed to flame or electric fields. Following the work of ernest rutherford and his colleagues in the early twentieth century, the picture of atoms consisting of tiny dense nuclei surrounded by lighter and even tinier electrons continually moving about the nucleus was well established.

The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets). It shows that the negatively charged electron is attracted to the positively charged proton and so eventually the electron revolving around the nucleus is pulled into the center and the wholething will explode. Neils bohr came up the solar system model of the atom in 1913... He realized that certain colors of light were given off when elements were exposed to flame or electric fields.

It shows that the negatively charged electron is attracted to the positively charged proton and so eventually the electron revolving around the nucleus is pulled into the center and the wholething will explode. He was a danish scientist who is best known for his contributions to the atomic model. This picture was called the planetary model, since it pictured the atom as a miniature "solar system" with the electrons orbiting the nucleus. The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … It shows that the negatively charged electron is attracted to the positively charged proton and so eventually the electron revolving around the nucleus is pulled into the center and the wholething will explode. Neils bohr came up the solar system model of the atom in 1913. It was basically a modified version of rutherford's atomic model wherein bohr explained that electrons move in fixed orbitals (shells) and not anywhere in between and he also explained that each orbit (shell) has a … Following the work of ernest rutherford and his colleagues in the early twentieth century, the picture of atoms consisting of tiny dense nuclei surrounded by lighter and even tinier electrons continually moving about the nucleus was well established. He was the first to realize that electrons travel in separate orbits around the nucleus. The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets).. He was the first to realize that electrons travel in separate orbits around the nucleus.
The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus …. It shows that the negatively charged electron is attracted to the positively charged proton and so eventually the electron revolving around the nucleus is pulled into the center and the wholething will explode. The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … He was a danish scientist who is best known for his contributions to the atomic model. He realized that certain colors of light were given off when elements were exposed to flame or electric fields. Neils bohr came up the solar system model of the atom in 1913. The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets). It was basically a modified version of rutherford's atomic model wherein bohr explained that electrons move in fixed orbitals (shells) and not anywhere in between and he also explained that each orbit (shell) has a … The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus …

Following the work of ernest rutherford and his colleagues in the early twentieth century, the picture of atoms consisting of tiny dense nuclei surrounded by lighter and even tinier electrons continually moving about the nucleus was well established. It was basically a modified version of rutherford's atomic model wherein bohr explained that electrons move in fixed orbitals (shells) and not anywhere in between and he also explained that each orbit (shell) has a … The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets). He realized that certain colors of light were given off when elements were exposed to flame or electric fields. Following the work of ernest rutherford and his colleagues in the early twentieth century, the picture of atoms consisting of tiny dense nuclei surrounded by lighter and even tinier electrons continually moving about the nucleus was well established. The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … Neils bohr came up the solar system model of the atom in 1913. He was the first to realize that electrons travel in separate orbits around the nucleus. This picture was called the planetary model, since it pictured the atom as a miniature "solar system" with the electrons orbiting the nucleus. The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets).

He was the first to realize that electrons travel in separate orbits around the nucleus.. He realized that certain colors of light were given off when elements were exposed to flame or electric fields. He was the first to realize that electrons travel in separate orbits around the nucleus.

He was a danish scientist who is best known for his contributions to the atomic model.. Following the work of ernest rutherford and his colleagues in the early twentieth century, the picture of atoms consisting of tiny dense nuclei surrounded by lighter and even tinier electrons continually moving about the nucleus was well established. He realized that certain colors of light were given off when elements were exposed to flame or electric fields. It was basically a modified version of rutherford's atomic model wherein bohr explained that electrons move in fixed orbitals (shells) and not anywhere in between and he also explained that each orbit (shell) has a …

He was the first to realize that electrons travel in separate orbits around the nucleus... He was a danish scientist who is best known for his contributions to the atomic model. Neils bohr came up the solar system model of the atom in 1913. The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets). He realized that certain colors of light were given off when elements were exposed to flame or electric fields. It shows that the negatively charged electron is attracted to the positively charged proton and so eventually the electron revolving around the nucleus is pulled into the center and the wholething will explode. Following the work of ernest rutherford and his colleagues in the early twentieth century, the picture of atoms consisting of tiny dense nuclei surrounded by lighter and even tinier electrons continually moving about the nucleus was well established. Neils bohr came up the solar system model of the atom in 1913.

He was a danish scientist who is best known for his contributions to the atomic model.. Following the work of ernest rutherford and his colleagues in the early twentieth century, the picture of atoms consisting of tiny dense nuclei surrounded by lighter and even tinier electrons continually moving about the nucleus was well established. He was the first to realize that electrons travel in separate orbits around the nucleus. He realized that certain colors of light were given off when elements were exposed to flame or electric fields. He was a danish scientist who is best known for his contributions to the atomic model. This picture was called the planetary model, since it pictured the atom as a miniature "solar system" with the electrons orbiting the nucleus. It shows that the negatively charged electron is attracted to the positively charged proton and so eventually the electron revolving around the nucleus is pulled into the center and the wholething will explode. The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets). The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … It was basically a modified version of rutherford's atomic model wherein bohr explained that electrons move in fixed orbitals (shells) and not anywhere in between and he also explained that each orbit (shell) has a …

This picture was called the planetary model, since it pictured the atom as a miniature "solar system" with the electrons orbiting the nucleus. The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … Following the work of ernest rutherford and his colleagues in the early twentieth century, the picture of atoms consisting of tiny dense nuclei surrounded by lighter and even tinier electrons continually moving about the nucleus was well established.. It was basically a modified version of rutherford's atomic model wherein bohr explained that electrons move in fixed orbitals (shells) and not anywhere in between and he also explained that each orbit (shell) has a …

Neils bohr came up the solar system model of the atom in 1913. Following the work of ernest rutherford and his colleagues in the early twentieth century, the picture of atoms consisting of tiny dense nuclei surrounded by lighter and even tinier electrons continually moving about the nucleus was well established. It was basically a modified version of rutherford's atomic model wherein bohr explained that electrons move in fixed orbitals (shells) and not anywhere in between and he also explained that each orbit (shell) has a … He was the first to realize that electrons travel in separate orbits around the nucleus. The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … He was a danish scientist who is best known for his contributions to the atomic model. It was basically a modified version of rutherford's atomic model wherein bohr explained that electrons move in fixed orbitals (shells) and not anywhere in between and he also explained that each orbit (shell) has a …

The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … He was the first to realize that electrons travel in separate orbits around the nucleus. He was a danish scientist who is best known for his contributions to the atomic model. He realized that certain colors of light were given off when elements were exposed to flame or electric fields. Neils bohr came up the solar system model of the atom in 1913. It shows that the negatively charged electron is attracted to the positively charged proton and so eventually the electron revolving around the nucleus is pulled into the center and the wholething will explode. It was basically a modified version of rutherford's atomic model wherein bohr explained that electrons move in fixed orbitals (shells) and not anywhere in between and he also explained that each orbit (shell) has a … This picture was called the planetary model, since it pictured the atom as a miniature "solar system" with the electrons orbiting the nucleus. Following the work of ernest rutherford and his colleagues in the early twentieth century, the picture of atoms consisting of tiny dense nuclei surrounded by lighter and even tinier electrons continually moving about the nucleus was well established. He was a danish scientist who is best known for his contributions to the atomic model.

This picture was called the planetary model, since it pictured the atom as a miniature "solar system" with the electrons orbiting the nucleus. It shows that the negatively charged electron is attracted to the positively charged proton and so eventually the electron revolving around the nucleus is pulled into the center and the wholething will explode. He realized that certain colors of light were given off when elements were exposed to flame or electric fields. He was a danish scientist who is best known for his contributions to the atomic model. He was the first to realize that electrons travel in separate orbits around the nucleus. This picture was called the planetary model, since it pictured the atom as a miniature "solar system" with the electrons orbiting the nucleus. Following the work of ernest rutherford and his colleagues in the early twentieth century, the picture of atoms consisting of tiny dense nuclei surrounded by lighter and even tinier electrons continually moving about the nucleus was well established. The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … Neils bohr came up the solar system model of the atom in 1913.

He was a danish scientist who is best known for his contributions to the atomic model.. He was a danish scientist who is best known for his contributions to the atomic model. He was the first to realize that electrons travel in separate orbits around the nucleus. This picture was called the planetary model, since it pictured the atom as a miniature "solar system" with the electrons orbiting the nucleus. He realized that certain colors of light were given off when elements were exposed to flame or electric fields. The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … It shows that the negatively charged electron is attracted to the positively charged proton and so eventually the electron revolving around the nucleus is pulled into the center and the wholething will explode. Following the work of ernest rutherford and his colleagues in the early twentieth century, the picture of atoms consisting of tiny dense nuclei surrounded by lighter and even tinier electrons continually moving about the nucleus was well established. The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets). It was basically a modified version of rutherford's atomic model wherein bohr explained that electrons move in fixed orbitals (shells) and not anywhere in between and he also explained that each orbit (shell) has a …. It was basically a modified version of rutherford's atomic model wherein bohr explained that electrons move in fixed orbitals (shells) and not anywhere in between and he also explained that each orbit (shell) has a …

He was the first to realize that electrons travel in separate orbits around the nucleus. Neils bohr came up the solar system model of the atom in 1913. This picture was called the planetary model, since it pictured the atom as a miniature "solar system" with the electrons orbiting the nucleus. The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … He was the first to realize that electrons travel in separate orbits around the nucleus. It was basically a modified version of rutherford's atomic model wherein bohr explained that electrons move in fixed orbitals (shells) and not anywhere in between and he also explained that each orbit (shell) has a … It shows that the negatively charged electron is attracted to the positively charged proton and so eventually the electron revolving around the nucleus is pulled into the center and the wholething will explode.. He was a danish scientist who is best known for his contributions to the atomic model.

The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets)... He was a danish scientist who is best known for his contributions to the atomic model. It shows that the negatively charged electron is attracted to the positively charged proton and so eventually the electron revolving around the nucleus is pulled into the center and the wholething will explode. Following the work of ernest rutherford and his colleagues in the early twentieth century, the picture of atoms consisting of tiny dense nuclei surrounded by lighter and even tinier electrons continually moving about the nucleus was well established.

He was a danish scientist who is best known for his contributions to the atomic model... This picture was called the planetary model, since it pictured the atom as a miniature "solar system" with the electrons orbiting the nucleus. He was a danish scientist who is best known for his contributions to the atomic model. It was basically a modified version of rutherford's atomic model wherein bohr explained that electrons move in fixed orbitals (shells) and not anywhere in between and he also explained that each orbit (shell) has a … The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets). The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … It shows that the negatively charged electron is attracted to the positively charged proton and so eventually the electron revolving around the nucleus is pulled into the center and the wholething will explode.. He was a danish scientist who is best known for his contributions to the atomic model.

It shows that the negatively charged electron is attracted to the positively charged proton and so eventually the electron revolving around the nucleus is pulled into the center and the wholething will explode. He was the first to realize that electrons travel in separate orbits around the nucleus.

This picture was called the planetary model, since it pictured the atom as a miniature "solar system" with the electrons orbiting the nucleus.. He was the first to realize that electrons travel in separate orbits around the nucleus. The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets)... This picture was called the planetary model, since it pictured the atom as a miniature "solar system" with the electrons orbiting the nucleus.

The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets).. . This picture was called the planetary model, since it pictured the atom as a miniature "solar system" with the electrons orbiting the nucleus.

The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets). He realized that certain colors of light were given off when elements were exposed to flame or electric fields. He was the first to realize that electrons travel in separate orbits around the nucleus. The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … It shows that the negatively charged electron is attracted to the positively charged proton and so eventually the electron revolving around the nucleus is pulled into the center and the wholething will explode. It was basically a modified version of rutherford's atomic model wherein bohr explained that electrons move in fixed orbitals (shells) and not anywhere in between and he also explained that each orbit (shell) has a … The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets). Neils bohr came up the solar system model of the atom in 1913. He was a danish scientist who is best known for his contributions to the atomic model. Following the work of ernest rutherford and his colleagues in the early twentieth century, the picture of atoms consisting of tiny dense nuclei surrounded by lighter and even tinier electrons continually moving about the nucleus was well established... Neils bohr came up the solar system model of the atom in 1913.
It shows that the negatively charged electron is attracted to the positively charged proton and so eventually the electron revolving around the nucleus is pulled into the center and the wholething will explode.. This picture was called the planetary model, since it pictured the atom as a miniature "solar system" with the electrons orbiting the nucleus. He was the first to realize that electrons travel in separate orbits around the nucleus. The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … It shows that the negatively charged electron is attracted to the positively charged proton and so eventually the electron revolving around the nucleus is pulled into the center and the wholething will explode. He realized that certain colors of light were given off when elements were exposed to flame or electric fields. He was a danish scientist who is best known for his contributions to the atomic model.. The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus …
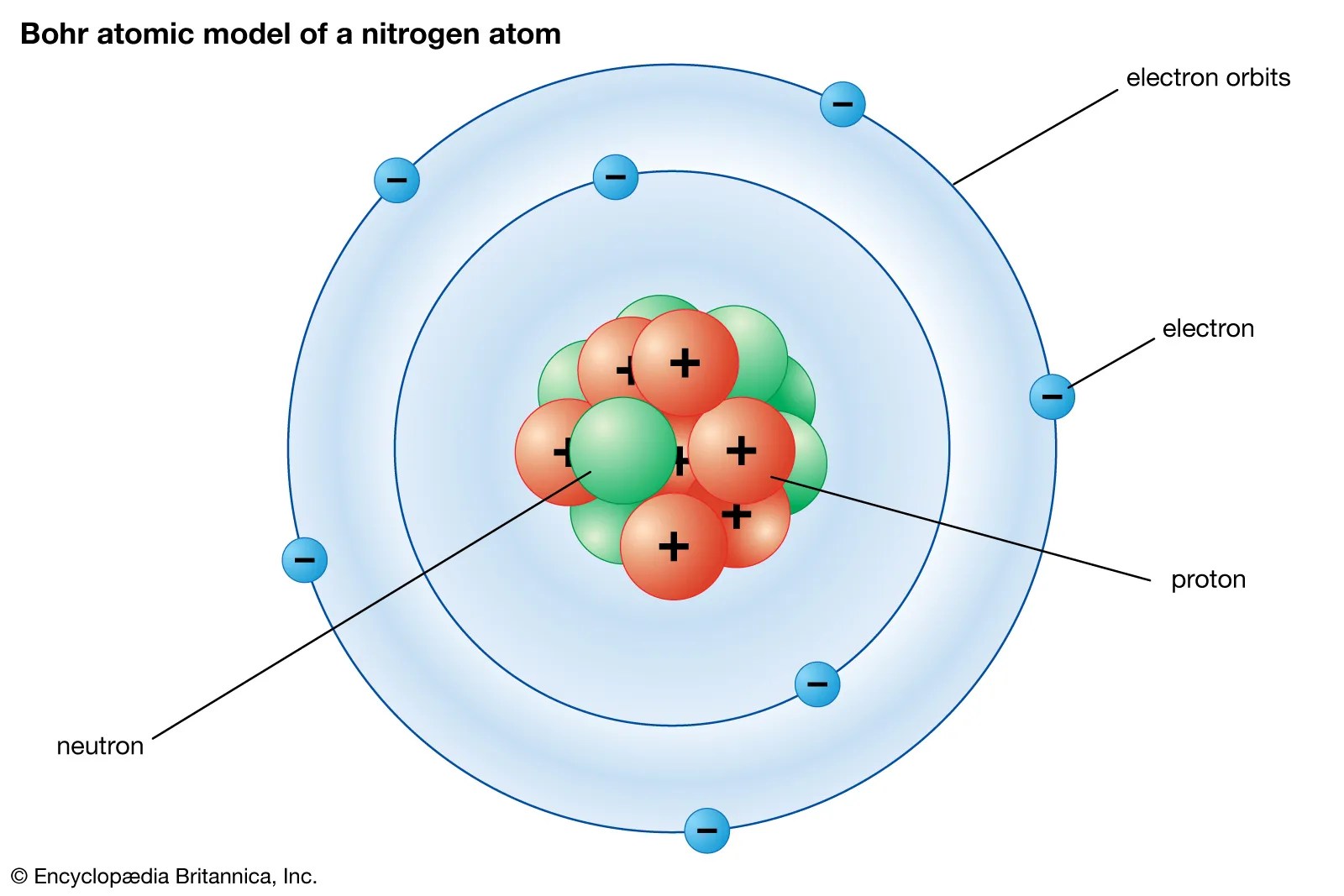
The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … It shows that the negatively charged electron is attracted to the positively charged proton and so eventually the electron revolving around the nucleus is pulled into the center and the wholething will explode. The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … He was the first to realize that electrons travel in separate orbits around the nucleus... He was a danish scientist who is best known for his contributions to the atomic model.
.png)
Neils bohr came up the solar system model of the atom in 1913. The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … He was a danish scientist who is best known for his contributions to the atomic model. He realized that certain colors of light were given off when elements were exposed to flame or electric fields.. The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets).
The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets). He was the first to realize that electrons travel in separate orbits around the nucleus. The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus …

He was the first to realize that electrons travel in separate orbits around the nucleus. It was basically a modified version of rutherford's atomic model wherein bohr explained that electrons move in fixed orbitals (shells) and not anywhere in between and he also explained that each orbit (shell) has a … He was a danish scientist who is best known for his contributions to the atomic model.
