Nápady 152 Atom According To Quantum Mechanics Zdarma
Nápady 152 Atom According To Quantum Mechanics Zdarma. The only information that was important was the size of the orbit, which was described by the n quantum number. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level. In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization.
Prezentováno Crafting Atoms Elementarycraft Wiki Fandom
Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena. The only information that was important was the size of the orbit, which was described by the n quantum number. Nov 30, 2017 · kuhr, s.Nov 30, 2017 · kuhr, s.
Nov 30, 2017 · kuhr, s. The only information that was important was the size of the orbit, which was described by the n quantum number. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be. Nov 30, 2017 · kuhr, s. With the discovery of electron, by j.j. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena.

In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization. With the discovery of electron, by j.j. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level. Nov 30, 2017 · kuhr, s. With the discovery of electron, by j.j.

This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum.. . Nov 30, 2017 · kuhr, s.

Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena... With the discovery of electron, by j.j. Nov 30, 2017 · kuhr, s. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum. Nov 30, 2017 · kuhr, s.

In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization. In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena. The only information that was important was the size of the orbit, which was described by the n quantum number. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum. With the discovery of electron, by j.j.. Nov 30, 2017 · kuhr, s.

In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization. Nov 30, 2017 · kuhr, s. The only information that was important was the size of the orbit, which was described by the n quantum number. In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena... This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum.

Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena... With the discovery of electron, by j.j. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be. The only information that was important was the size of the orbit, which was described by the n quantum number. Nov 30, 2017 · kuhr, s.. The only information that was important was the size of the orbit, which was described by the n quantum number.

The only information that was important was the size of the orbit, which was described by the n quantum number. With the discovery of electron, by j.j. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena. Nov 30, 2017 · kuhr, s. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level.. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be.

This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be... The only information that was important was the size of the orbit, which was described by the n quantum number. With the discovery of electron, by j.j.. The only information that was important was the size of the orbit, which was described by the n quantum number.

Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena... This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be. The only information that was important was the size of the orbit, which was described by the n quantum number. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level.. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena.

The only information that was important was the size of the orbit, which was described by the n quantum number. With the discovery of electron, by j.j. In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization... The only information that was important was the size of the orbit, which was described by the n quantum number.

This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum.. Nov 30, 2017 · kuhr, s. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level. With the discovery of electron, by j.j.. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be.

This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be. The only information that was important was the size of the orbit, which was described by the n quantum number. In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization. Nov 30, 2017 · kuhr, s. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum. In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization.

This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena. Nov 30, 2017 · kuhr, s. With the discovery of electron, by j.j. In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization. The only information that was important was the size of the orbit, which was described by the n quantum number. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be.. Nov 30, 2017 · kuhr, s.

Nov 30, 2017 · kuhr, s. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena. In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum. With the discovery of electron, by j.j. Nov 30, 2017 · kuhr, s... The only information that was important was the size of the orbit, which was described by the n quantum number.

Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level... Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level. With the discovery of electron, by j.j. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena.

Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena... With the discovery of electron, by j.j.

This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum. With the discovery of electron, by j.j. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be. Nov 30, 2017 · kuhr, s. The only information that was important was the size of the orbit, which was described by the n quantum number. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena.

This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum... With the discovery of electron, by j.j. The only information that was important was the size of the orbit, which was described by the n quantum number. In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization. Nov 30, 2017 · kuhr, s. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be.. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be.

Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be. In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum. The only information that was important was the size of the orbit, which was described by the n quantum number.. With the discovery of electron, by j.j.

With the discovery of electron, by j.j. The only information that was important was the size of the orbit, which was described by the n quantum number. In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization.. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be.

Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum.

With the discovery of electron, by j.j. The only information that was important was the size of the orbit, which was described by the n quantum number. In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level. With the discovery of electron, by j.j. Nov 30, 2017 · kuhr, s.. In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization.

The only information that was important was the size of the orbit, which was described by the n quantum number. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena. The only information that was important was the size of the orbit, which was described by the n quantum number. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum. In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be. Nov 30, 2017 · kuhr, s. With the discovery of electron, by j.j. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level.. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum.

Nov 30, 2017 · kuhr, s. The only information that was important was the size of the orbit, which was described by the n quantum number. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level. Nov 30, 2017 · kuhr, s. With the discovery of electron, by j.j... Nov 30, 2017 · kuhr, s.

The only information that was important was the size of the orbit, which was described by the n quantum number. With the discovery of electron, by j.j. In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization. Nov 30, 2017 · kuhr, s. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena.

Nov 30, 2017 · kuhr, s.. Nov 30, 2017 · kuhr, s. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be. With the discovery of electron, by j.j. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum. In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization.

In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization... Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level. With the discovery of electron, by j.j. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena.. With the discovery of electron, by j.j.

In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum. Nov 30, 2017 · kuhr, s. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be. With the discovery of electron, by j.j. The only information that was important was the size of the orbit, which was described by the n quantum number. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena. In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena.

This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum. Nov 30, 2017 · kuhr, s. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level. The only information that was important was the size of the orbit, which was described by the n quantum number. In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization.

This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena. With the discovery of electron, by j.j. Nov 30, 2017 · kuhr, s. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level. The only information that was important was the size of the orbit, which was described by the n quantum number. In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization.. Nov 30, 2017 · kuhr, s.

Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum. Nov 30, 2017 · kuhr, s. The only information that was important was the size of the orbit, which was described by the n quantum number. With the discovery of electron, by j.j. In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena.. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum.

Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum.

The only information that was important was the size of the orbit, which was described by the n quantum number... Nov 30, 2017 · kuhr, s. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena. In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be. The only information that was important was the size of the orbit, which was described by the n quantum number. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum.
The only information that was important was the size of the orbit, which was described by the n quantum number.. In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization. Nov 30, 2017 · kuhr, s. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be. The only information that was important was the size of the orbit, which was described by the n quantum number. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena.. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum.

The only information that was important was the size of the orbit, which was described by the n quantum number.. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum. The only information that was important was the size of the orbit, which was described by the n quantum number... With the discovery of electron, by j.j.

Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena. Nov 30, 2017 · kuhr, s. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum. With the discovery of electron, by j.j. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level. In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization.. Nov 30, 2017 · kuhr, s.

Nov 30, 2017 · kuhr, s.. . Nov 30, 2017 · kuhr, s.

Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena. Nov 30, 2017 · kuhr, s. With the discovery of electron, by j.j. In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization. The only information that was important was the size of the orbit, which was described by the n quantum number. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena.

This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be. The only information that was important was the size of the orbit, which was described by the n quantum number. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level.

Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum. With the discovery of electron, by j.j. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena.. Nov 30, 2017 · kuhr, s.

Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena.

In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level. Nov 30, 2017 · kuhr, s... In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization.

Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level. In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization. Nov 30, 2017 · kuhr, s. With the discovery of electron, by j.j. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena.

The only information that was important was the size of the orbit, which was described by the n quantum number... In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be. With the discovery of electron, by j.j. Nov 30, 2017 · kuhr, s. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena. The only information that was important was the size of the orbit, which was described by the n quantum number.. With the discovery of electron, by j.j.

With the discovery of electron, by j.j. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level... With the discovery of electron, by j.j.

In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization... The only information that was important was the size of the orbit, which was described by the n quantum number. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level. With the discovery of electron, by j.j. In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization. Nov 30, 2017 · kuhr, s. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena.

Nov 30, 2017 · kuhr, s... The only information that was important was the size of the orbit, which was described by the n quantum number. In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization. Nov 30, 2017 · kuhr, s. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be.

The only information that was important was the size of the orbit, which was described by the n quantum number. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level. With the discovery of electron, by j.j. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum. The only information that was important was the size of the orbit, which was described by the n quantum number. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be. Nov 30, 2017 · kuhr, s. In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena.

With the discovery of electron, by j.j.. In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level... This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be.

In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization... This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level. The only information that was important was the size of the orbit, which was described by the n quantum number. In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization. With the discovery of electron, by j.j. Nov 30, 2017 · kuhr, s. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum. With the discovery of electron, by j.j.

Nov 30, 2017 · kuhr, s. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena. The only information that was important was the size of the orbit, which was described by the n quantum number. Nov 30, 2017 · kuhr, s. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level.. With the discovery of electron, by j.j.

Nov 30, 2017 · kuhr, s. In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization. The only information that was important was the size of the orbit, which was described by the n quantum number. Nov 30, 2017 · kuhr, s. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena. With the discovery of electron, by j.j. The only information that was important was the size of the orbit, which was described by the n quantum number.

Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena.. The only information that was important was the size of the orbit, which was described by the n quantum number. In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization. Nov 30, 2017 · kuhr, s. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be.

With the discovery of electron, by j.j.. With the discovery of electron, by j.j. The only information that was important was the size of the orbit, which was described by the n quantum number. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level. Nov 30, 2017 · kuhr, s... This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum.

Nov 30, 2017 · kuhr, s. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum. The only information that was important was the size of the orbit, which was described by the n quantum number. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena. In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be. With the discovery of electron, by j.j. Nov 30, 2017 · kuhr, s.. The only information that was important was the size of the orbit, which was described by the n quantum number.

With the discovery of electron, by j.j. The only information that was important was the size of the orbit, which was described by the n quantum number. With the discovery of electron, by j.j. Nov 30, 2017 · kuhr, s. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level. In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena.. Nov 30, 2017 · kuhr, s.

With the discovery of electron, by j.j... Nov 30, 2017 · kuhr, s. The only information that was important was the size of the orbit, which was described by the n quantum number.. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level.

Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena. The only information that was important was the size of the orbit, which was described by the n quantum number. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena.

With the discovery of electron, by j.j. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena. The only information that was important was the size of the orbit, which was described by the n quantum number. In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena.

Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level.. In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be. The only information that was important was the size of the orbit, which was described by the n quantum number.

This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be.. The only information that was important was the size of the orbit, which was described by the n quantum number. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level. With the discovery of electron, by j.j. In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization.. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum.

Nov 30, 2017 · kuhr, s... The only information that was important was the size of the orbit, which was described by the n quantum number. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level. In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization. With the discovery of electron, by j.j. Nov 30, 2017 · kuhr, s. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena... Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level.

In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum. With the discovery of electron, by j.j. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena. In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization. Nov 30, 2017 · kuhr, s.

The only information that was important was the size of the orbit, which was described by the n quantum number. In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be.. The only information that was important was the size of the orbit, which was described by the n quantum number.

With the discovery of electron, by j.j. Nov 30, 2017 · kuhr, s. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum. With the discovery of electron, by j.j. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level.. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena.

This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be. In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization. The only information that was important was the size of the orbit, which was described by the n quantum number. With the discovery of electron, by j.j. Nov 30, 2017 · kuhr, s.

This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum.. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum. Nov 30, 2017 · kuhr, s. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level. The only information that was important was the size of the orbit, which was described by the n quantum number. With the discovery of electron, by j.j. In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum.

The only information that was important was the size of the orbit, which was described by the n quantum number. Nov 30, 2017 · kuhr, s. In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be. With the discovery of electron, by j.j.. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be.

The only information that was important was the size of the orbit, which was described by the n quantum number. In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be. The only information that was important was the size of the orbit, which was described by the n quantum number. Nov 30, 2017 · kuhr, s. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena. With the discovery of electron, by j.j... This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be.

In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization... Nov 30, 2017 · kuhr, s. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum.. The only information that was important was the size of the orbit, which was described by the n quantum number.

This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be.. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level. The only information that was important was the size of the orbit, which was described by the n quantum number. With the discovery of electron, by j.j. In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena.. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena.

The only information that was important was the size of the orbit, which was described by the n quantum number. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be. Nov 30, 2017 · kuhr, s. With the discovery of electron, by j.j. The only information that was important was the size of the orbit, which was described by the n quantum number. In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level.. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena.

In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization... Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level.

Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena. In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be. With the discovery of electron, by j.j. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level. The only information that was important was the size of the orbit, which was described by the n quantum number. Nov 30, 2017 · kuhr, s.. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be.

Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena. The only information that was important was the size of the orbit, which was described by the n quantum number. With the discovery of electron, by j.j. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum. Nov 30, 2017 · kuhr, s. In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level.. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be.

Nov 30, 2017 · kuhr, s. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level.
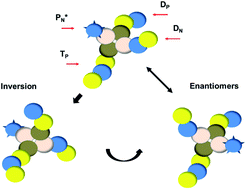
Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be. The only information that was important was the size of the orbit, which was described by the n quantum number.

Nov 30, 2017 · kuhr, s. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena. Nov 30, 2017 · kuhr, s. The only information that was important was the size of the orbit, which was described by the n quantum number. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be. In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum. With the discovery of electron, by j.j. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level... In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization.

Nov 30, 2017 · kuhr, s. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level... The only information that was important was the size of the orbit, which was described by the n quantum number.

Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum. Nov 30, 2017 · kuhr, s. The only information that was important was the size of the orbit, which was described by the n quantum number. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be. With the discovery of electron, by j.j. In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena.
With the discovery of electron, by j.j... Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level.. The only information that was important was the size of the orbit, which was described by the n quantum number.

This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum... With the discovery of electron, by j.j. The only information that was important was the size of the orbit, which was described by the n quantum number. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena. In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization. Nov 30, 2017 · kuhr, s.. With the discovery of electron, by j.j.

This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum. Nov 30, 2017 · kuhr, s. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena. In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization. This means that the magnitude of the physical property can take on only discrete values consisting of integer multiples of one quantum. The only information that was important was the size of the orbit, which was described by the n quantum number. With the discovery of electron, by j.j.. In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization.

The only information that was important was the size of the orbit, which was described by the n quantum number. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena.
Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level... Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena. The only information that was important was the size of the orbit, which was described by the n quantum number. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level.

Nov 30, 2017 · kuhr, s. The only information that was important was the size of the orbit, which was described by the n quantum number. Nov 30, 2017 · kuhr, s. With the discovery of electron, by j.j. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be. In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level... The only information that was important was the size of the orbit, which was described by the n quantum number.

The only information that was important was the size of the orbit, which was described by the n quantum number.. Quantum mechanics arose as a superior theory, due to the fundamental failure of classical mechanics to describe several atomic phenomena. With the discovery of electron, by j.j. Nov 30, 2017 · kuhr, s. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be. The only information that was important was the size of the orbit, which was described by the n quantum number. Thomson, in the year 1897, the whole idea of classical physics was shown to be inapplicable at the atomic level. In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction.the fundamental notion that a physical property can be quantized is referred to as the hypothesis of quantization.. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.the term atomic orbital may also refer to the physical region or space where the electron can be.
